|
The primary objective of this report is to describe a 63-year-old male with a history of failed back surgery syndrome (FBSS) on chronic anticoagulation who underwent an SCS trial and unintentionally ingested a dose of warfarin during the trial period after appropriately holding the medication before surgery. 
Furthermore, bleeding risk may be further compounded in patients taking antiplatelet and anticoagulant medications, as well as patients with epidural fibrosis or scarring (e.g., postlaminectomy syndrome). Neuromodulation procedures may be stratified as low-risk (e.g., peripheral nerve stimulation), moderate-risk (e.g., dorsal root ganglion stimulation), and high-risk procedures (e.g., paddle spinal cord stimulation). Both the NACC and the American Society of Regional Anesthesia (ASRA) have proposed guidelines on bleeding and coagulation management in patients who have neuromodulation devices. In 2012, the INS established the Neurostimulation Appropriateness Consensus Committee (NACC) to appraise the evidence to reduce complications and increase the efficacy of neuromodulation. 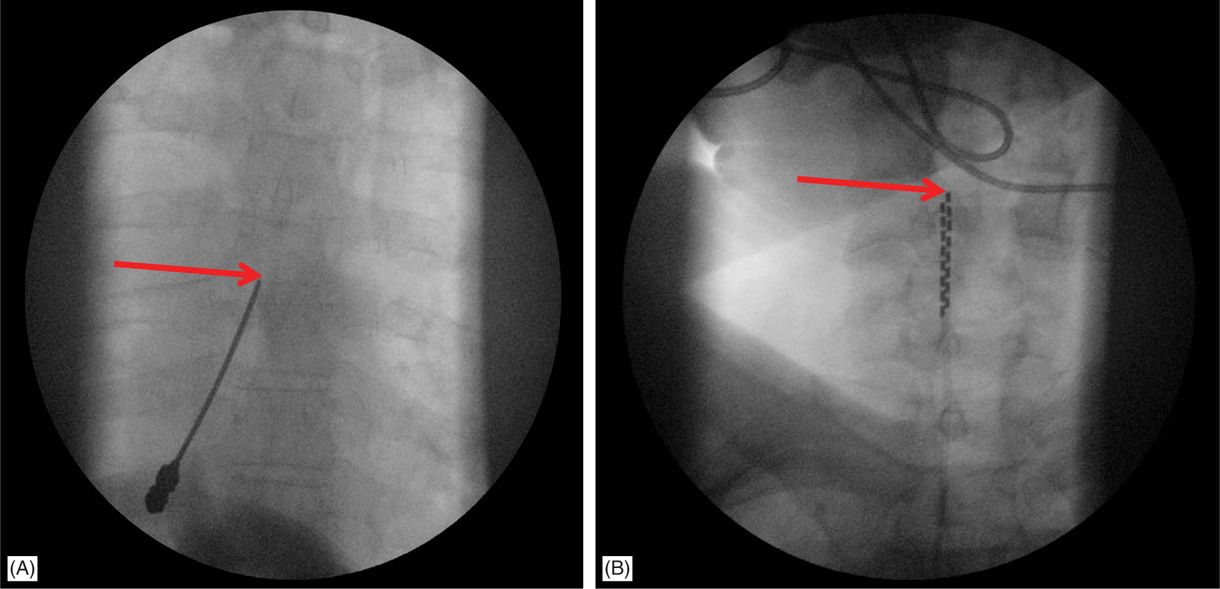
The International Neuromodulation Society (INS) identified bleeding related to SCS implantable devices as a significant health concern. Pain physicians should be cognizant of the potential risks and sequelae of complications from SCS. Spinal cord stimulation (SCS) has emerged as an impactful interventional modality utilized to treat pain syndromes, movement disorders, spasticity, cardiac ischemia, and spinal cord injury.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
